Volume, mass and density - formulas
Are you struggling with volume, mass and density calculations? We are here to end it once and for all. We are here to make your volume, mass and density calculations as easy as possible. Imagine you can make any calculation of volume, mass or density without any problems. You do not have to imagine it anymore. Just read this article carefully.
What will you find in this article? First of all, 3 calculations formulas. First for volume calculation, second for mass calculation and third for density calculation. What’s next? The step by step explanation with practical examples. This is not another theoretical text from which you will learn nothing. We prepared this article as a compendium with the practical knowledge ready to use immediately after reading it. So, without any further ado, let’s move on. It’s time to finally learn how to calculate volume, mass and density.
How to find volume with density and mass
The practical part we will start by explaining to you how to find volume with density and mass. As we said in the introduction, the base thing you need to know to make a calculation like this is a formula. So what is the volume formula? It looks as follows:

- Volume = Mass / Density
The volume formula can be also written in another way, using the symbols of these 3 units instead of their full names. Have a look:
- V = m / ρ
A quick explanation of all these symbols to make it clear: V is the volume, m is the mass and ρ is the density.
So as you can see, to find the volume with density and mass, you need to divide the mass by the density. It is only one calculation, nothing more. You have to admit that it doesn’t look complicated.
So if you know the volume formula, we can show you how to find volume with density and mass in practice. An example is down below.
For instance, the average block of concrete weighs about 25 kilograms. In turn, the average density of the concrete is about 2400 kilograms per cubic meter. So let’s write the data:
- m = 25 kg;
- ρ = 2400 kg/m3.
Now we can figure the data into the volume formula. It will look as follows:
- V = 25 kg / 2400 kg/m3
And now have a look at the calculations:
- V = 25 kg / 2400 kg/m3 = 0.0104166667 m3
So as you can see, the volume is equal to 0.0104166667 cubic meters. So now the data looks like this:
- m = 25 kg;
- ρ = 2400 kg/m3;
- V = 0.0104166667 m3.
If you do not need such an accurate result, you can round it off, for instance, to two decimal places. Then the volume will be equal to 0.01 cubic meters.
The volume formula and volume calculations are behind us, let’s move on to another measurement – the mass.
How to find mass with density and volume
To know how to find mass with density and volume you need to know the mass formula first. The formulas are the basic thing in all calculations in this article. So what is the mass formula? Have a look:
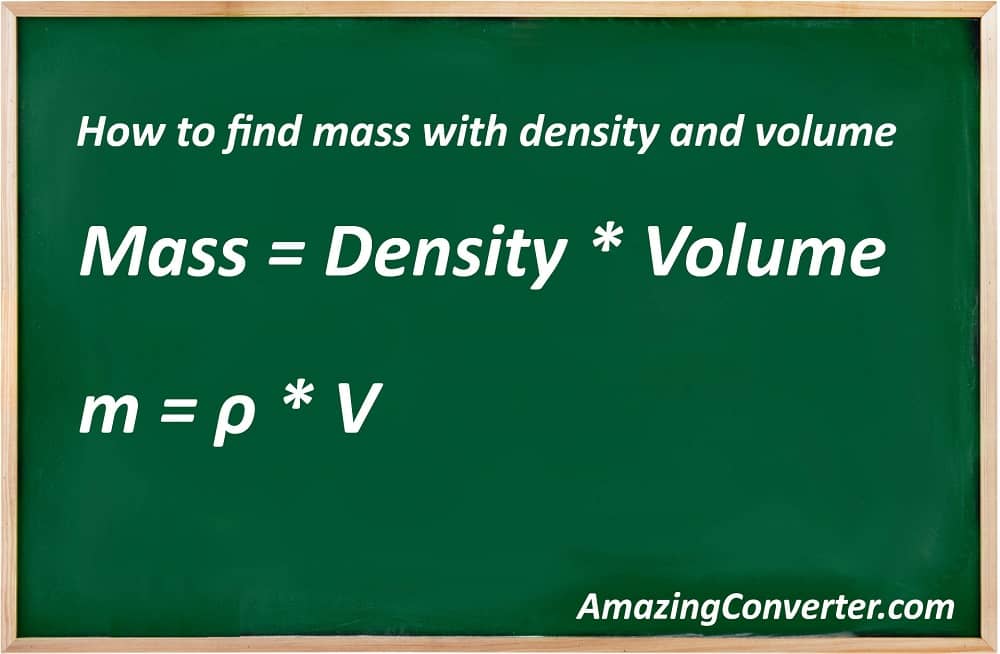
- Mass = Density * Volume
It is also possible to write it using the symbols, not full names. It will look like this:
- m = ρ * V
The symbols we have explained in the previous part, so we won’t repeat it here.
The mass formula is a little bit different, because the calculation which you need to do is multiplying, not dividing. So, to sum up – how to calculate mass using density and volume? You need to multiply two numbers, the number of the density and the number of the volume.
So let’s have a look at an example and learn how to find mass with density and volume.
For instance, the density of water is equal to 997 kg/m3. Let’s calculate it for 2 liters. The data looks like this:
- ρ = 997 kg/m3;
- V = 2 liters = 0.002 m3.
As you can see, we turn liters into cubic meters. Why? Because to calculate the mass properly, you need the same units in the density and volume. Otherwise the result will be incorrect.
So how does the mass equation look?
- m = 997 kg/m3 * 0.002 m3
And here are the calculations of the mass:
- m = 997 kg/m3 * 0.002 m3 = 1.994 kg
As you can see, the result is equal to 1.994 kilograms. Now the data is:
- ρ = 997 kg/m3;
- V = 2 liters = 0.002 m3;
- m = 1.994 kg.
We want to draw your attention to one issue – 2 liters is not equal to 2 kilograms. Of course, 1.994 kilograms are approximately 2 kilograms after rounding off to the round number, but it is not an exact result then.
We have also another example for you. A quick reminder before it. How do you find mass with volume? To find mass with volume you also need density. Then you have to multiply the volume by the density. Now we want to show you calculations made for another unit of volume. Will they look the same as for calculating mass with volume? Let’s check it. Have a look:
How do you find mass with volume and ML?
You need to multiply the volume ML by the density. It will look like this:
- Mass = Volume ML * Density
Now let’s see it in practice. The data with the volume ML is given down below. The density is given for oil.
- V = 200 ml;
- ρ = 950 kg/m3.
As you can see, the density is in another unit of volume than the unit using ML, so it is needed to change it. We will make a conversion from kg/m3 to g/mL. Have a look:
- ρ = 950 kg/m3 = 0.95 g/mL
Now we can start calculations. The equation to find mass with volume and ML looks as follows:
- m = 200 mL * 0.95 g/mL = 0.19 g
So the result is 0.19 grams. This time the volume is not equal to mass too. It means 200 mL is not equal to 200 milligrams, but only to 190 milligrams (because 0.19 g = 190 mg).
Before we move on to the density calculation, we want to answer the three questions often asked by you, which closely concerns the topic of finding mass.
How to find weight from density? To find the weight from density, you need to multiply the density by the volume. After this calculation you will get the result in the unit of mass, as a weight.
How do you find the mass of a density function? The density function looks as follows: ρ = m/V. To find the mass of a density function you need to transform the function by multiplying by the volume. It will look like this: ρV = m. So you can find the mass of a density function by transforming it. Now you need to multiply the density by the volume to get the mass.
How do you find mass with only volume? To find mass you need volume and density, only the volume is not sufficient. Why? Because the formula for mass looks as follows: m = ρV. If you know which substance you are going to calculate mass, you can check the density table.
How to find density with mass and volume
The last calculation is the calculation for density with mass and volume. As in the two previous examples, the first thing you need to know is a formula. What is the density formula? Let’s see:
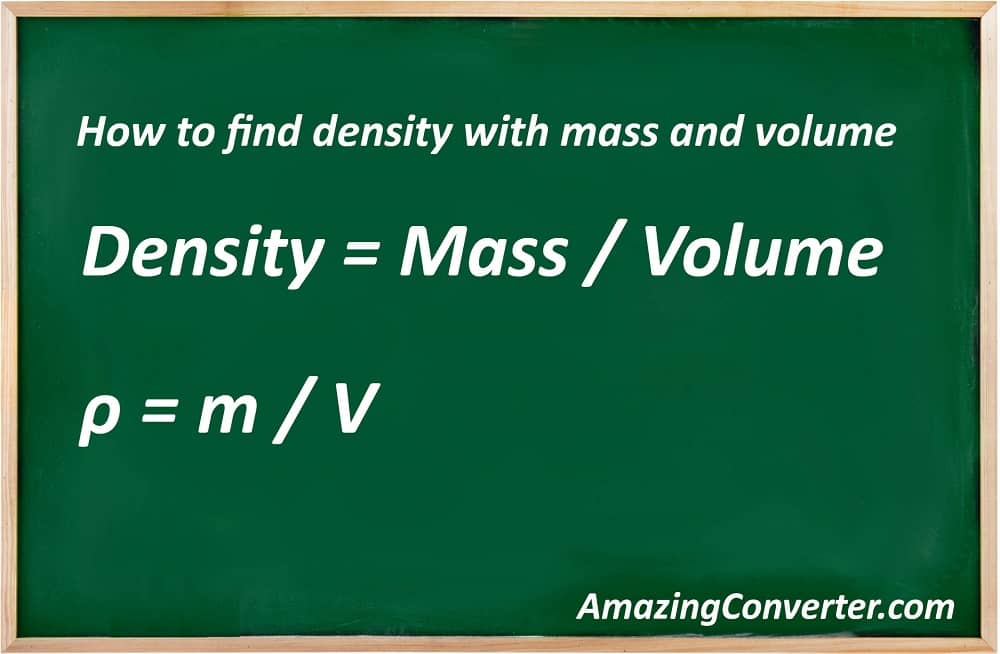
- Density = Mass / Volume
The density formula with the use of symbols instead of full names of measurements looks like this:
- ρ = m / V
For example for example, the density of water donverter
So how to find density with mass and volume?
To find the density you need to divide the mass by the volume. Let’s see it in practice.
For instance, calculate the density of the mass equals to 5 kg and volume equals to 3 liters. The data looks as follows:
- m = 5 kg;
- V = 0.003 m3.
So the equation for the density with mass and volume looks like this:
- ρ = 5 kg / 0.003 m3
And here are the calculations to find the density with mass and volume:
- ρ = 5 kg / 0.003 m3 = 1666.66667 kg/m3
So the result is 1666.66667 kg/m3. The data now looks as follows:
- m = 5 kg;
- V = 0.003 m3;
- ρ = 1666.66667 kg/m3.
The density value you can also round off, for instance, to two decimal places. Then the result will be equal to 1666.67 kg/m3.
We are sure that if you have carefully read the whole article, you know now how to calculate volume, mass and density with three different formulas – volume formula, mass formula and density formula. So it is time to use this knowledge in practice. Make your own volume, mass and density calculations according to the instructions, finally without any problems.









